Difference between revisions of "Designing XOR Gates - two campus approach"
(→Missouri Western XOR Biological Design) |
(→Missouri Western XOR Biological Design) |
||
| Line 21: | Line 21: | ||
| − | [[Image: | + | [[Image:XOR AI2.JPG]]<br> |
| − | [[Image: | + | [[Image:XOR AI1.JPG]]<br> |
== Davidson Ampicillin Communication: time delayed cell growth == | == Davidson Ampicillin Communication: time delayed cell growth == | ||
Revision as of 21:15, 12 June 2008
Contents
Davidson XOR Biological Design
Here is an idea Malcolm had.
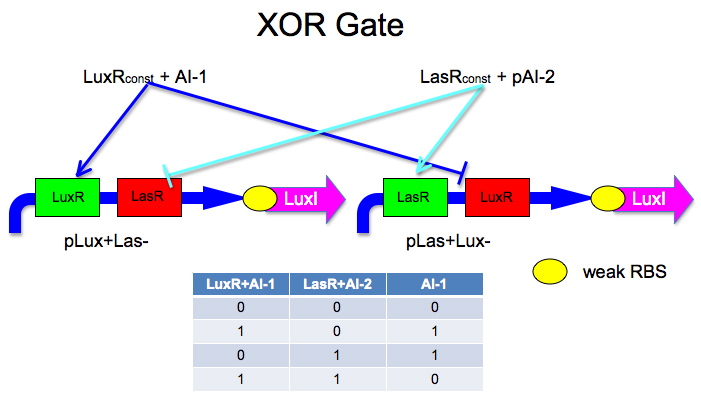
The idea is to have two mirrored halves of the system. One is regulated by AI-1 and the other by AI-2. There is a potential problem in that the Lux half is more likely to get positive feedback than the Las half. This MAY not be a problem because 0/0 is leaky so we put a weak RBS to minimize leaky protein production. Also, if we add AI-2 and AI-1 is produced by leak, then the entire system shuts down. The repressor site is located between -35 and -10 of the promoter. The activator binding site is upstream of -35. This has been documented by Egland and Greenberg
Missouri Western XOR Biological Design
Here is an idea Todd had.
MWSU: The protein in the diagram should be LacI, not LacIq. If you want to, you can indicate LacI X86+I12.
Also, the output gene should be LuxI, not LuxS. With LuxS, the second cell has to have the Las system in place of the Lux system.
JB/AMC
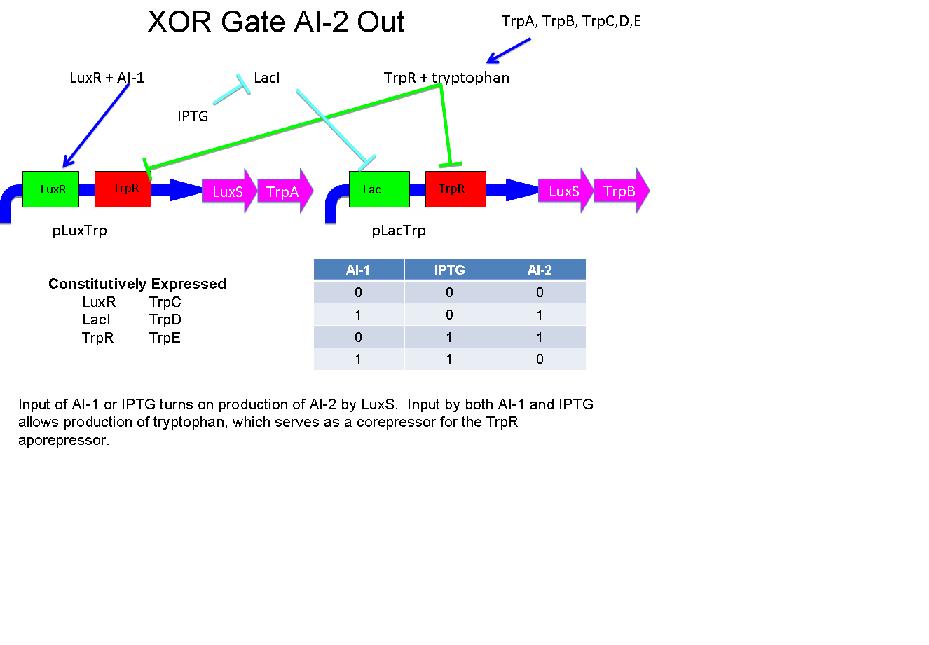
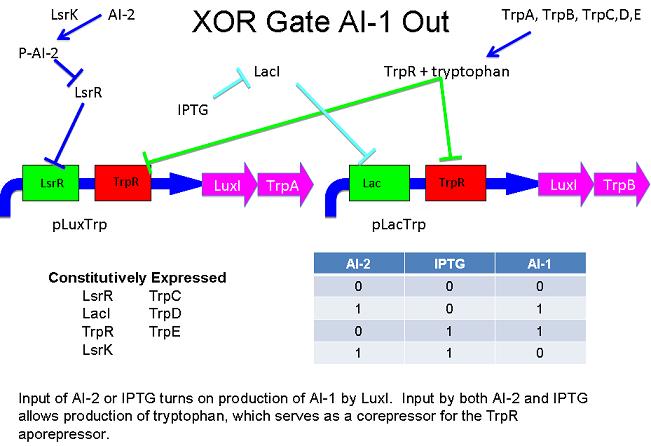
XOR Based on Tryptophan Anabolism and the TrpR Repressor
Davidson Ampicillin Communication: time delayed cell growth
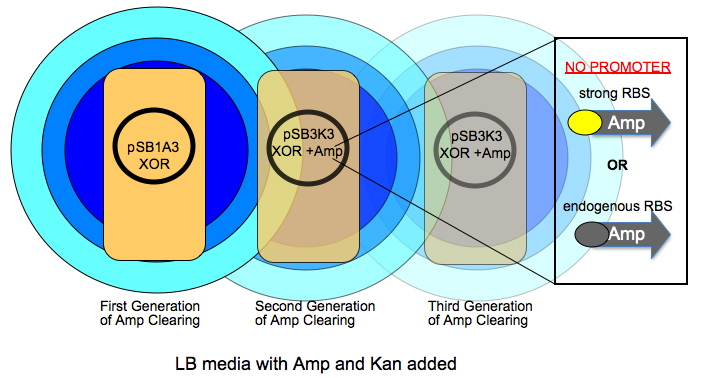
The very first colony has a high copy number plasmid that is Amp and Kan resistant (R). As this colony grows over time, it will digest ampicillin in an increasingly larger circle shown by radiating circles of blue. The subsequent colonies have a lower copy number plasmid and have a promoter-less version of AmpR in addition to the XOR construct. The AmpR coding will have either its native RBS or one we give it.
Davidson Growth Layouts
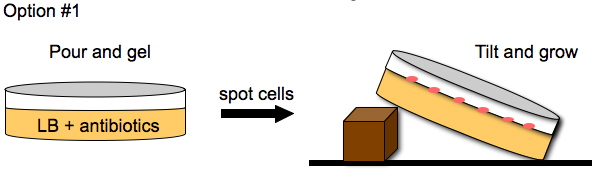
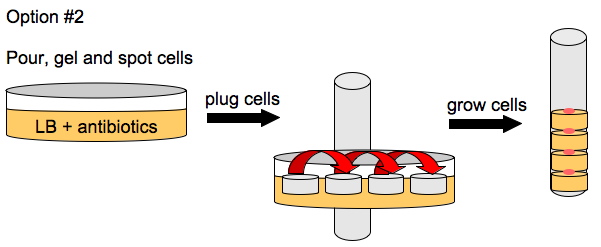
To enhance the unidirectional flow of AmpR, we could either grow the cells on a slant or create a vertical stack of agar plugs. The thickness of the plug would be determined by the thickness of the plates we pour. This may or may not help with the diffusion of AmpR but it is easier to do than microfluidics.