Will Notebook2
Wideloache 15:40, 23 January 2009 (EST)
Today I
Wideloache 14:25, 22 January 2009 (EST)
After two days, there was still no growth in the 2 liquid cultures (Amp/Gen and Gen). There were 5-6 colonies on each of the LB + Gen plates (one was at 10 ug/mL and one was at 20 ug/mL). I streaked one of the colonies onto an LB + Gen plate at 20 ug/mL to save in the fridge. I also grew up 3 colonies in different liquid culture conditions at 37C: LB+Gen, LB+Gen+Amp, and LB+Amp. This should give me information on whether both plasmids are in the cells. Cells that have the genomics insertion only should only grow in LB+Gen. Cells that have both of the insertion plasmids should grow in all 3 conditions.
I also miniprepped and digested J61002-J23100 with EcoRI and PstI. This should give me pSB1A2 vector with RFP cut out. I ran this digestion on a 0.8% gel and obtained the following result:
Wideloache 17:14, 20 January 2009 (EST)
On the 16th, I found that my negative control transformation had tons of colonies (almost a lawn). The two experimental plates had equal numbers of colonies. I didn't bother testing these colonies because they were almost certainly not the desired constructs. I'm going to have to redo the transformation with a new vector digestion. Therefore, today I grew up J61002-J23100 in LB + Amp. Tomorrow I will miniprep and digest the plasmid in preparation for another transformation. It is possible that the MP of J61002-J23100 was bad, since I didn't have the second expected band when I did the E/P digestion on the 15th.
I also made new Gentamicin plates and LB + Gen liquid culture. I made two different plate concentrations: 10 ug/mL and 20 ug/mL. My liquid culture was at 15 ug/mL which was the concentration in which Chris said to grow up the colonies containing pInt80 and pG80ko. [Note: Last semester I had used 20 ug/mL, which was the literature working concentration. When I did the procedure last semester I figured that the concentration difference wouldn't be a problem, but since I'm trouble shooting now, I made new liquid culture at the correct concentration.]
I picked HB101 +pInt80-649 +pG80ko-K091206 out of the -80. I streaked them out of the LB Gen plates at both concentrations. I will keep track of the growth of these cells to see if there is a difference in growth time. If the 10 ug/mL plates have faster growing cells, this might suggest that the gentamicin concentration of 20 ug/mL was too high. I grew these plates at 37.
I grew the HB101 +pInt80-649 +pG80ko-K091206 cells in LB + gen and LB +gen+amp liquid culture at 37. Theoretically these cells should grow in both tubes.
At this point, most of these variable growth conditions are just to try to wrap my head around what could be going on inside the cells based on their response to different growth conditions.
Wideloache 15:45, 15 January 2009 (EST)
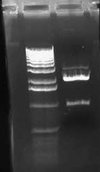
I ran my digestion of J61002-J23100 (E/P) on a 0.8% gel and got the following result:
I expected to see a band at around 900, which would have been the RFP insert, but since the vector was fairly faint as well, it probably just wasn't bright enough to see. I excised the band at around 2000 bps which should be pSB1A2 vector cut with E/P.
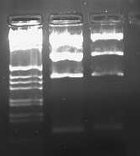
I also ran my digestions of LacIX86-pLux' and LacII12X86-pLuxLas on a 2.0% gel and got the following result.
This was what I expected to see, and I excised the lowest band on each gel (expected size was 93 bps).
I did a gel purification of all 3 excised gel fragments, and then I ligated pSB1A2 with each of the pLux promoter parts. I also did a negative control ligation of pSB1A2 + H20. I expect to have the unsuccessful ligation be red, making the verification of successful ligations easier. I plated on LB Amp plates after transforming into JM109 cells.
Wideloache 20:36, 13 January 2009 (EST)
Today was my first day back in the lab of the semester. The first thing that I needed to do was to get the two pLux promoters that had been synthesized by GeneArt into pSB1A2. I had done an (E/P) digestion of pLux' and pLuxLas last semester that seemed to be successful. However, when I tried to transform into pSB1A2, I had a low colony yield. I decided to make a fresh stock of pSB1A2 digested with E/P. I used the part J61002-J23100. Cutting this with E/P woudl result in a pSB1A2 vector with a RFP expression insert. In this way I could screen out unsuccessful ligations. In addition to running this digestion O/N, I also digested pLux' and pLuxLas with E/P from the minipreps of LacIX86-pLux' and LacII12X86-pLuxLas.
Make GenR plates (15 ug/mL) Miniprep pLas' and pLasLux Get GFP plasmid to put 4 promoters in front of.